1. Introduction
Water splitting is an efficient method for producing clean hydrogen, which can be utilized as an alternative energy source for traditional fossil fuels [1-3]. Water electrolysis involves the anodic oxygen evolution reaction (OER) and the cathodic hydrogen evolution reaction (HER) [4-6]. The slow kinetics of the OER at the anode always leads to a high overpotential, which decreases the efficiency of water splitting and results in a low hydrogen production rate on the cathode. Although some noble-metal materials such as ruthenium/iridium oxides (RuO2/IrO2) have been proven to be efficient catalysts for the OER [7-9], their large-scale commercialization is limited due to their high cost and low production. Accordingly, the development of efficient and cheap OER electrocatalysts for hydrogen production by water electrolysis has become a topic of great interest [10].
In recent years, the development of 3d-transition-metal-based materials for the OER has gained interest because of their earth abundance, low cost, and high efficiency [11-13]. In addition, layered double hydroxides (LDHs) are considered promising candidates owing to their high activity as well as defect-rich t2g orbitals in the MO6-x center, which presumably accelerate the adsorption reactions of OH- anions [14-16]. However, the low utilization efficiency of the active sites and poor electronic conductivity restrict the OER performance of these materials, and many approaches have been proposed to further improve their performance. One of the available methods for regulating the electronic structure and enhancing their OER performance is doping with other elements. To date, studies on the incorporation of trivalent elements, such as V, Cr, and Mn into the LDH nanostructure have been reported; this method imparts excellent OER capability to the pristine material because of the tuned electronic structure in LDHs [17-19].
In a new approach, it was found that metalloid elements can significantly change the electronic configuration of transition metals by inducing different bonding processes that combine ionic, covalent, or metallic interactions [20-22]. Because of their moderate electronegativity, which ranges from 1.9 to 2.1, metalloids can accept electrons from transition metals and transport them to neighboring oxygen atoms in the LDHs. This allows the targeted generation of electron-enriched or electron-depleted local regions on the surface of LDHs, which may be beneficial for maximizing the number of active sites for electrocatalytic water splitting. Furthermore, electronic coupling interactions between metal d-orbitals and metalloid sp-orbitals can shift the center of the d-orbital from the Fermi energy level, leading to improved electrical conductivity for LDHs. Finally, the incorporation of metalloids into transition metal hydroxides can further strengthen electrocatalytic stability, since the metalloids can form covalent bonds with the surrounding transition metals in linear, planar, and 3D structures, thus resulting in impressive physicochemical resistance [23-25].
In this study, we show that metalloid incorporation into transition metal LDHs is a promising strategy for designing highly efficient and commercial electrocatalysts for alkaline OER. Tellurium (Te) was selected as a promising metalloid for modulating the catalytic properties of nickel-cobalt LDH (NiCo LDH), where the electrical conductivity and number of active sites could be simultaneously enhanced. To the best of our knowledge, metalloid incorporation for tuning the electrocatalytic properties of LDHs has rarely been studied despite its high application potential. In order to fill this gap, Te was introduced into NiCo LDH grown on three-dimensional (3D) porous nickel foam (NF) by a facile solvothermal method in this study. The resulting material showed superior OER performance due to the optimized electronic structure resulting from the introduced Te and the conductive NF as the current collector. The optimum Te amount introduced into the NiCo LDH is discussed in terms of the electrocatalytic OER performance.
2. Experimental Procedure
2.1 Synthesis of NiCo LDH and ŽćTe-NiCo LDHs
NiCo LDH and Te-incorporated NiCo LDHs were synthesized by a simple hydrothermal method [26]. NFs were treated with acetone and then with 3 M HCl solution for 10 min using an ultrasonication cleaner. The cleaned NFs were then dried overnight. All chemicals were purchased from Kojundo Chemical and used directly without further purification. To synthesize the NiCo LDH, the as-prepared NFs were immersed in 20 mL of deionized (DI) water. Then, 1.00 mg of Co (II) nitrate hexahydrate (Co(NO3)2┬Ę6H2O) was dissolved in the solution at room temperature. Sonication was performed for 10 min to completely dissolve the Co precursor in the solution. The resulting solutions were transferred to a Teflonlined autoclave and hydrothermally reacted at 180 ┬░C for 5 h. After several washes with deionized water, the NiCo LDH samples were dried in a vacuum at 60 ┬░C overnight. The synthesis method for the ŽćTe-NiCo LDHs was the same as that for NiCo-LDH, except that tellurium tetrachloride (TeCl4) was added to the solution before the hydrothermal reaction. The stoichiometric ratios (Žć) of Te to Co in the ŽćTe-CoNi LDHs were 0.2, 0.4, 0.6 and 0.8.
2.2. Characterizations
Microstructural images of each sample were observed by field-emission scanning electron microscopy (FE-SEM; model S4800; Hitachi) with energy-dispersive X-ray (EDX) spectrometry. X-ray diffraction (XRD) was performed using a D/MAX-2500/PC (Rigaku) diffractometer at 40 kV and 100 mA with Cu-K╬▒ radiation (╬╗ = 0.15418 nm). The electrochemical properties of the catalysts in 1 M KOH were tested using a three-electrode electrochemical cell controlled by an electrochemical workstation (Autolab PGSTAT; Metrohm), in which the catalyst grown on NF was used directly as the working electrode. Prior to the measurements, the electrolyte (1 M KOH, pH Ōēł 13.7) was purged for approximately 10 min with O2, and the working electrodes were sealed on all edges with a custom-made acrylate adhesive, leaving working surface area of 0.25 cm2 untreated. A graphite rod and Hg|Hg2SO4 were used as the counter and reference electrodes, respectively. A clear glass titration vessel was used as the testing cell. The distance between the working and reference electrodes was approximately 1 cm. Linear sweep voltammetry (LSV) was performed at a scan rate of 0.5 mV sŌłÆ1 in the range from 1.23 to 1.9 V vs. a reversible hydrogen electrode (RHE). The applied potentials were calibrated against the RHE, and all polarization curves were iR-corrected. Electrochemical impedance spectroscopy (EIS) measurements were conducted over the frequency range of 0.1ŌĆō100 kHz at 1.45 VRHE with a sinusoidal amplitude of 5 mV.
To quantitatively evaluate the catalytic activity of the NiCo LDH and ŽćTe-NiCo LDH samples, we estimated the doublelayer capacitance (Cdl) using a cyclic voltammetry (CV) method in a non-Faradaic region where the current generated from the electrical double layer charging is essentially related to electrochemical surface area (ECSA). A CV test was conducted in an O2-saturated 1 M KOH solution to estimate the Cdl at non-Faradaic overpotentials. The CV measurements were performed at various scan rates (20, 40, 60, 80, 100, and 120 mV/s). The difference in current density between the anodic and cathodic sweeps (JanodicŌĆōJcathodic) at the middle of potential range was plotted as a function of the scan rate, where the slope has a linear relationship with twice the Cdl of the catalyst.
3. Results and Discussion
NiCo LDH exhibits poor electrical conductivity, which impedes facile charge transfer between the surface of the catalyst and the adsorbed reactant. Following Te incorporation, the electrical properties of the ŽćTe-NiCo LDHs significantly improved, so that they could easily accept electrons in the hydroxyl ions, significantly accelerating the water oxidation kinetics. Te is preferentially incorporated at the edge sites of the transition metal, that is, a real catalytic active site in LDHs, where strong covalent p-d hybridization occurs with a highly polarized local electronic structure. The latter significantly accelerates the electrocatalytic OER activity of ŽćTe-NiCo LDHs [27].
NiCo LDH and ŽćTe-NiCo LDH nanosheets were synthesized by direct growth on the NF using tellurium chloride and cobalt nitrate via a hydrothermal reaction at 180 ┬░C for 5 h. Figure 1 shows highly magnified FE-SEM images of the asgrown NiCo LDH and ŽćTe-NiCo (Žć = 0.4, 0.6, 0.8) LDH nanosheets. The surface morphology of the 0.2Te-NiCo LDH sample resembled an intermediate nanosheet structure with NiCo LDH and 0.4Te-NiCo LDH, as shown in Fig. S1. Figure 1 further shows that large-scale highly interconnected and aligned nanosheet structures grew vertically on the skeletons of the NF with a uniform morphology and dense loading, forming an ordered and 3D network with a highly open and interstitial structure. A homogeneous nanosheet structure on the 3D macroporous NF was clearly observed for the NiCo LDH and ŽćTe-NiCo (Žć = 0.2, 0.4, 0.6) LDHs.
However, when the ratio of the Te precursor vs. Co exceeded 0.6, the homogeneous growth of nanosheets on the NF was suppressed, leading to the formation of irregular and collapsed nanostructures on the surface of the NF. A further increase in the ratio led to the formation of a highly aggregated particle morphology with a large area of bare NF, uncovered by the grown material. Figure S2 shows a low-magnification FE-SEM image of the NiCo LDH and ŽćTe-NiCo (Žć = 0.2, 0.4, 0.6, 0.8) LDH nanosheet arrays on the NF. The 3D macroscopically porous structure of the NF was well maintained, and the surface of the entire NF became rougher with increasing Te content, as shown in Fig. S2(d,e).
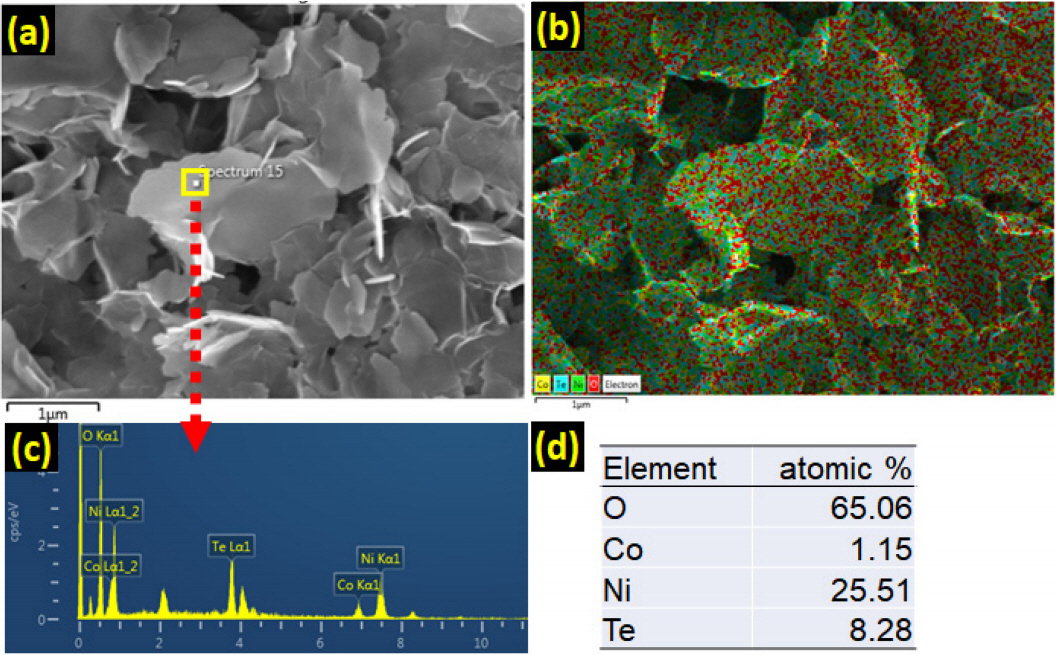
Energy-dispersive X-ray (EDX) spectroscopy analysis of the as-prepared ŽćTe-NiCo LDHs revealed the existence of Co, Ni, O and Te. Figure 2 shows an (a) FE-SEM image, (b) EDX mapping image, (c) EDX spectrum, and (d) atomic composition of the 0.6Te-NiCo LDH sample. The EDX mapping image of O, Co, Ni, and Te elements in 0.6Te-NiCo LDH indicates all elements are homogeneously distributed. Quantitative EDX analysis showed that the atomic ratio of O:Co:Ni:Te in the 0.6Te-NiCo LDH sample was 65.06:1.15: 25.51:8.28. Figure S3 shows the variation in the atomic composition of the NiCo LDH and ŽćTe-NiCo (Žć = 0.2, 0.4, 0.6, 0.8) LDHs, as calculated by EDX analysis.
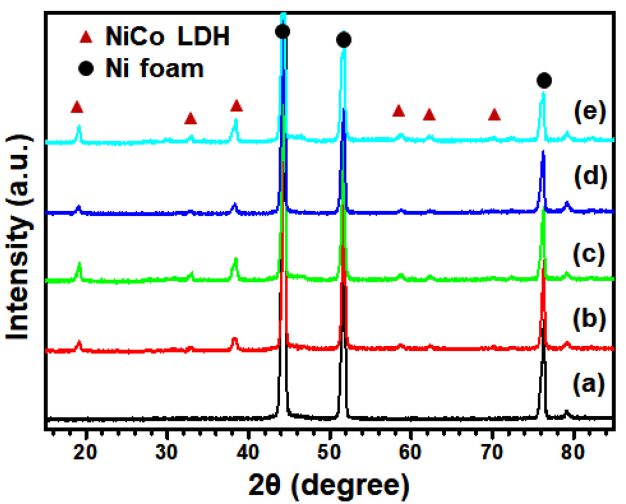
Typical XRD analysis also confirmed the formation of the NiCo LDH and ŽćTe-NiCo crystal structures as shown in Fig 3. The strong diffraction peaks near 44 ┬║ and 51 ┬║ could be assigned to Ni metal in the NF substrate. The XRD patterns revealed the crystal structure and phase purity of the NiCo LDH nanosheets. Except for the peaks from the NF substrate, all other detectable diffraction peaks at low 2╬Ė angles could be matched well with the hydrotalcite-like LDH phase [28]. Prominent diffraction peaks for the layered nickel-cobalt hydroxide were observed at 19 ┬║, 33 ┬║ and 38 ┬║, which were seen more clearly in the enlarged XRD profile depicted in Fig. S4. Notably, a secondary phase related to Te (Ni2.6Te2) was observed at 28 ┬║, 30 ┬║, 34 ┬║ and 46 ┬║ in the XRD pattern of the 0.8Te-NiCo LDH sample, indicating that a very high Te content is not desirable for synthesizing Te-incorporated transition metal hydroxide nanosheets on the NF substrate [29].
The electrocatalytic activities of the NiCo LDH and ŽćTe-NiCo LDH samples were tested in alkaline media (1 M KOH aqueous solution) using a three-electrode system. LSV was performed on the sample at a scan rate of 0.5 mV/s. NF substrates, on which self-supported catalysts were grown, were directly used as working electrodes, while a rotating disk electrode (RDE) was employed for testing powder samples such as RuO2. The RDE was continuously rotated at 2,000 rpm to remove the bubbles generated during measurement. All measured potentials were iR-compensated and then referenced to a reversible hydrogen electrode (RHE). Notably, ŽćTe-NiCo LDHs exhibited enhanced catalytic activity for water oxidation as compared to the NiCo LDH sample. The overpotential (╬Ę) required to transmit a current density of 10 mA/cm2 (╬Ę10) is conventionally used as a standard to compare electrocatalytic OER performance [30].
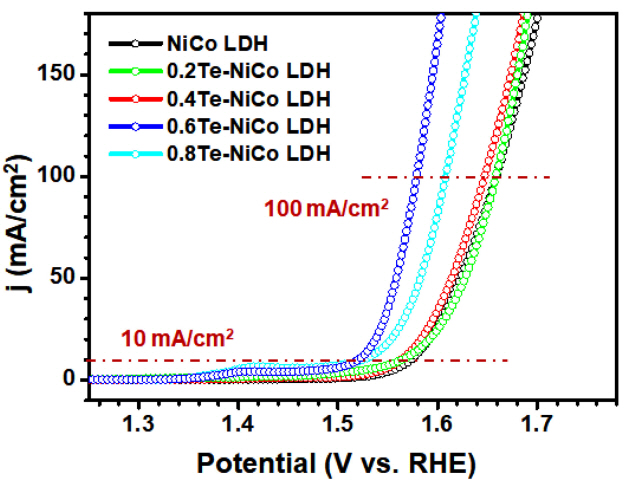
Figure 4 shows LSV curves obtained at a scan rate of 0.5 mV/s for evaluating electrocatalytic OER properties of the NiCo LDH and ŽćTe-NiCo LDHs. The ╬Ę10 value substantially decreased when Te was introduced into NiCo LDH. The 0.6Te-NiCo LDH sample required an ╬Ę10 of 290 mV, while ╬Ę10 for the NiCo LDH was about 350 mV as shown in Fig 4. Importantly, an overpotential of only 330 mV was required for the 0.6Te-NiCo LDH to generate a high current density of 100 mA/cm2, which can be useful in practical electrolysis applications. The amount of incorporated Te involved in the reaction was found to play a crucial role in improving catalytic activity. In terms of overpotential, the Te content in the 0.6Te-NiCo LDH resulted in the best catalytic activity for water oxidation. However, further increasing the Te content (0.8Te-NiCo LDH) significantly deteriorated the catalytic activity, presumably due to poorly grown nanosheets on the NF substrate, as demonstrated by the FE-SEM images in Fig 1(d) and Fig. S2(e).
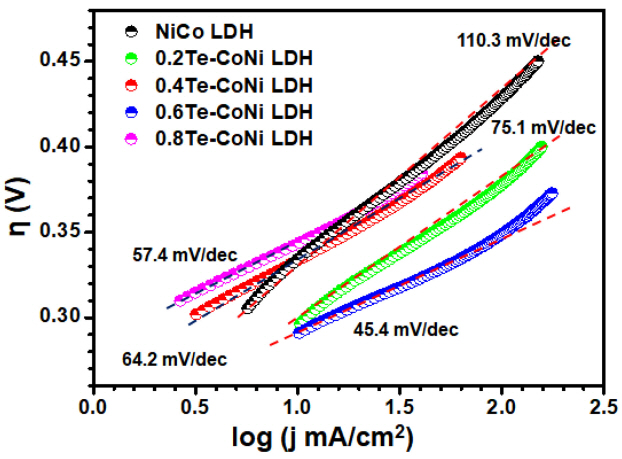
Tafel plots of the samples were derived from the measured LSV curves based on the Tafel equation (╬Ę = b ├Ś logj + a), where ╬Ę is the overpotential, j is the current density, and b is the Tafel slope. The Tafel slopes of the NiCo LDH and ŽćTe-NiCo LDHs were calculated and are shown in Fig 5. The measured Tafel slopes for the NiCo LDH were 110.3, and those for the ŽćTe-NiCo LDHs were 75.1, 64.2, 45.4, and 57.4 mV/dec for Žć values of 0.2, 0.4, 0.6, and 0.8, respectively. The 0.6Te-NiCo LDH exhibited a much smaller Tafel slope (45.4 mV/dec) compared to the NiCo LDH (110.3 mV/dec), highlighting the potential use of Te-NiCo LDHs as industrial electrolyzer because a smaller Tafel slope is desirable for reducing power losses [31].
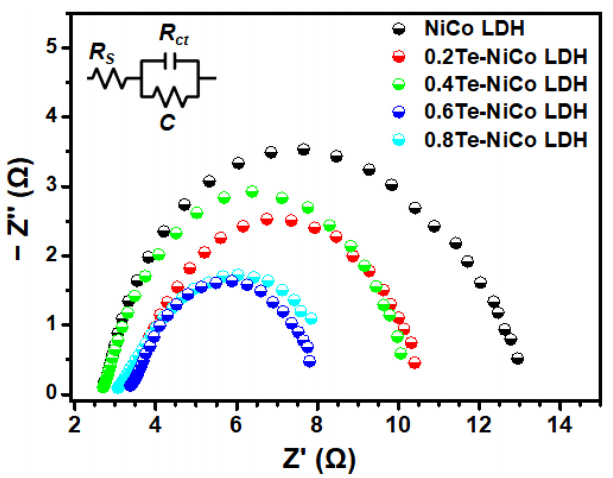
The charge transfer resistance (Rct) between electrocatalysts and electrolytes can be determined from the semicircle diameter in the high-frequency region of the Nyquist plot (Z╩╣ vs. ŌĆōZ╩╣╩╣) measured by electrochemical impedance spectroscopy (EIS). Here, a smaller diameter generally represents a lower Rct value. Figure 6 represents the EIS data for the samples, where ŽćTe-NiCo LDHs (8 ŌöĆ 10 Ōä”) showed an Rct value obviously lower than that of the NiCo LDH sample (~13 Ōä”). Notably, the 0.6Te-NiCo LDH exhibited the minimum Rct (~8 Ōä”), implying the significant role of Te incorporation in facilitating charge transfer from the catalyst surface to the adsorbed chemical reactants. A further increase in the Te content (0.8Te-NiCo LDH) increased the charge transfer resistance, which may be related to the poorly grown nanosheets on the NF substrate, which reinforces the results obtained from the LSV curves and Tafel slopes.
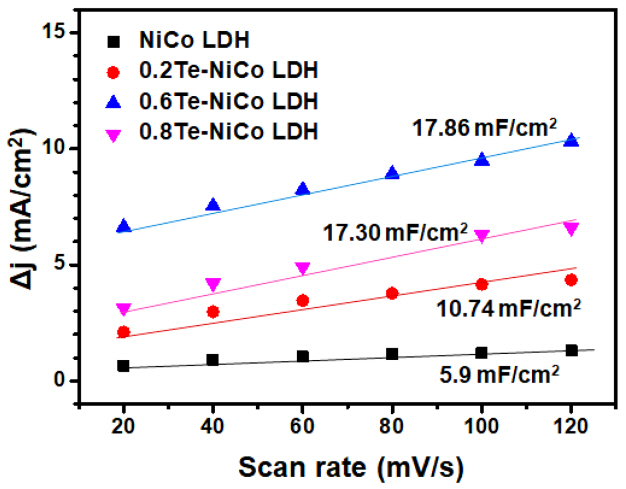
The CV test was conducted in an O2-saturated 1 M KOH solution to estimate the Cdl at non-Faradaic overpotentials. CV measurements were performed at various scan rates (20, 40, 60, 80, 100, and 120 mV/s) as shown in Fig S5. The difference in current density between the anodic and cathodic sweeps (JanodicŌĆōJcathodic) in the middle of the potential range was plotted as a function of the scan rate, where the slope has a linear relationship with twice the Cdl of the catalyst. The Cdl was calculated to be 17.86 mF for 0.6Te-NiCo LDH sample, while the NiCo LDH, 0.2Te-NiCo LDH and 0.2Te-NiCo LDH samples exhibited much lower Cdl values of 5., 10.74 and 17.30 mF/cm2, respectively as shown in Fig 7.
4. Conclusions
In summary, Te-incorporated nickel cobalt LDHs were developed as highly efficient and low-cost electrocatalysts for water oxidation under alkaline conditions. Pristine NiCo LDH and ŽćTe-NiCo (Žć = 0.2, 0.4, 0.6, and 0.8) LDH nanosheets were successfully grown via a hydrothermal reaction at 180 ┬░C for 5 h. A homogeneous nanosheet structure on the NF was clearly observed for the NiCo LDH and ŽćTe-NiCo (Žć = 0.2, 0.4, 0.6) LDHs. Irregular and collapsed nanostructures were found on the surface of the NF when the Te ratio (Žć) exceeded 0.6. The XRD patterns revealed that the crystal structures of the NiCo LDH and ŽćTe-NiCo could be indexed to the hydrotalcite-like LDH phase. Secondary phases were found in the 0.8Te-NiCo LDH sample, indicating that a high Te content is inappropriate for synthesizing ŽćTe-NiCo LDHs. The 0.6Te-NiCo LHD electrocatalyst exhibited current densities of 10 and 100 mA/ cm2 at overpotentials of only 290 and 330 mV, respectively, with a very small Tafel slope of 45.48 mV/dec in an alkaline medium. Moreover, the 0.6Te-NiCo LDH exhibited a minimum Rct (~8 Ōä”) with respect to the NiCo LDH sample (~13 Ōä”), implying the significant role of Te incorporation in facilitating charge transfer from the catalyst surface to the adsorbed chemical reactants. The double layer capacitance Cdl was calculated to be 17.86 mF for the 0.6Te-NiCo LDH compared to the value of 5.9 for the pristine NiCo LDH. These results demonstrate that ŽćTe-NiCo LDHs are promising electrocatalysts for water oxidation, with the optimum Te content (Žć) being 0.6.